有机物异丙醇(C3H8O)可由生物质转化得到,催化异丙醇脱水制取高值化学品丙烯(C3H6)的工业化技术已引起人们的关注,其主要反应如下:Ⅰ.C3H8O(g)⇌C3H6(g)+H2O(g)△H1=+52kJ/molⅡ.2C3H6(g)=C6H12(g)△H2=-97kJ/mol在1350℃下,恒容密闭容器中的反应体系内水蒸气浓度与反应时间关系如下表: 反应时间/μs 0 4 8 12 t 20 H2O浓度/ppm 0 2440 3200 3600 4000 4100 (1)4~8μs内,v(C3H8O)= ____ ppm/μs。(2)t ____ 16(填“>”、“<”或“=”)。(3)在恒温恒容密闭容器中,反应Ⅰ、Ⅱ均达到平衡的判据是 ____ (双选)。A.H2O(g)的浓度不变B.混合气体密度不变C.n(C3H6)=n(C6H12)D.v正(H2O)=v逆(C3H8O)(4)当2v正(C3H6)=v逆(C6H12)时,反应Ⅱ处于 ____ (填写“正向进行”、“逆向进行”或“化学平衡状态”)。假设某恒容容器内只发生反应Ⅱ(仅存在C3H6和C6H12两种物质),若要使反应Ⅱ达到平衡后,向正反应方向移动,可以改变的条件是 ____ (列举两条)。如图为反应Ⅰ、Ⅱ达到平衡时1gK与温度的关系曲线。6-|||-4-|||-曲线M-|||-甲-|||-2 曲线N 丙-|||-0 于-|||--2 戊-|||-4-|||-150 200 250 300 350 400-|||-温度/℃(5)根据反应热效应,判断曲线M对应反应 ____ (填写“Ⅰ”或“Ⅱ”),判断依据为 ____ 。(6)在350℃恒容平衡体系中充入少量水蒸气时,此刻反应Ⅰ的状态最有可能对应图中的 ____ 点(填“甲”、“乙”、“丙”、“丁”或“戊”),判断依据是 ____ 。(7)350℃时,在密闭恒容容器中加入一定量的C3H8O,体系达到平衡后,测得C6H12的物质的量浓度为xmol/L,C3H6的物质的量浓度为 ____ mol/L(用含x的代数式表示)。
有机物异丙醇(C3H8O)可由生物质转化得到,催化异丙醇脱水制取高值化学品丙烯(C3H6)的工业化技术已引起人们的关注,其主要反应如下:Ⅰ.C3H8O(g)⇌C3H6(g)+H2O(g)△H1=+52kJ/mol
Ⅱ.2C3H6(g)=C6H12(g)△H2=-97kJ/mol
在1350℃下,恒容密闭容器中的反应体系内水蒸气浓度与反应时间关系如下表:
| 反应时间/μs | 0 | 4 | 8 | 12 | t | 20 |
| H2O浓度/ppm | 0 | 2440 | 3200 | 3600 | 4000 | 4100 |
(2)t ____ 16(填“>”、“<”或“=”)。
(3)在恒温恒容密闭容器中,反应Ⅰ、Ⅱ均达到平衡的判据是 ____ (双选)。
A.H2O(g)的浓度不变
B.混合气体密度不变
C.n(C3H6)=n(C6H12)
D.v正(H2O)=v逆(C3H8O)
(4)当2v正(C3H6)=v逆(C6H12)时,反应Ⅱ处于 ____ (填写“正向进行”、“逆向进行”或“化学平衡状态”)。假设某恒容容器内只发生反应Ⅱ(仅存在C3H6和C6H12两种物质),若要使反应Ⅱ达到平衡后,向正反应方向移动,可以改变的条件是 ____ (列举两条)。
如图为反应Ⅰ、Ⅱ达到平衡时1gK与温度的关系曲线。
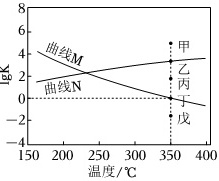
(5)根据反应热效应,判断曲线M对应反应 ____ (填写“Ⅰ”或“Ⅱ”),判断依据为 ____ 。
(6)在350℃恒容平衡体系中充入少量水蒸气时,此刻反应Ⅰ的状态最有可能对应图中的 ____ 点(填“甲”、“乙”、“丙”、“丁”或“戊”),判断依据是 ____ 。
(7)350℃时,在密闭恒容容器中加入一定量的C3H8O,体系达到平衡后,测得C6H12的物质的量浓度为xmol/L,C3H6的物质的量浓度为 ____ mol/L(用含x的代数式表示)。
参考答案与解析:
-
相关试题
-
[习 越]-|||-1.根据碳是四价,氢是一价,氧是二价,把下列分子式写成任何一种可能的构造式(用-|||-简写式表示)。-|||-(1)C3H8 (2)C3H8O (3)C4H10(4)C3H6
-
[习 越]-|||-1.根据碳是四价,氢是一价,氧是二价,把下列分子式写成任何一种可能的构造式(用-|||-简写式表示)。-|||-(1)C3H8 (2)C3H
- 查看答案
-
丙烯是重要的化工原料,利用丙烷制备丙烯通常有三种方法。方法一:丙烷直接脱氢法(1)丙烷直接脱氢的主要反应为C3H8(g)⇌C3H6(g)+H2(g),△H1=+129.80kJ•mol-1。600℃、
-
丙烯是重要的化工原料,利用丙烷制备丙烯通常有三种方法。方法一:丙烷直接脱氢法(1)丙烷直接脱氢的主要反应为C3H8(g)⇌C3H6(g)+H2(g),△H1=+
- 查看答案
-
工业上用乙苯脱氢制苯乙烯-|||-._(6)(H)_(5)(C)_(2)(H)_(5)(g)=!=!= (C)_(6)(H)_(5)(C)_(2)(H)_(3)(g)+(H)_(2)(g) .-|||
-
工业上用乙苯脱氢制苯乙烯-|||-._(6)(H)_(5)(C)_(2)(H)_(5)(g)=!=!= (C)_(6)(H)_(5)(C)_(2)(H)_(3)
- 查看答案
-
6.工业上制取氢气时涉及的一个重要反应是:-|||-(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)-|||-已知:-|||-①C(s)+1/2 O2(g)=CO
-
6.工业上制取氢气时涉及的一个重要反应是:-|||-(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)-|||-已知:-||
- 查看答案
-
在一定T、p下,乙苯脱氢反应:C6H5?C2H5(g)→C6H5?C2H3(g)+H2(g)反应达平衡后再加入一定量的H2O(g),则此反应的Ky();乙苯的平衡转化率α()。
-
[单选题]在一定T、p下,乙苯脱氢反应:C6H5?C2H5(g)→C6H5?C2H3(g)+H2(g)反应达平衡后再加入一定量的H2O(g),则此反应的Ky()
- 查看答案
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+(H)_(2)(g)-|||-Delta
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+
- 查看答案
-
(log )_(k)^-1-|||-(H)_(3)(g)+5(O)_(2)(g)=4NO(g)+6(H)_(2)O(g)-|||-Delta (H)_(3)=-907.3kJcdot mo(l)^-1
-
(log )_(k)^-1-|||-(H)_(3)(g)+5(O)_(2)(g)=4NO(g)+6(H)_(2)O(g)-|||-Delta (H)_(3)=-
- 查看答案
-
2.完成下列反应式。-|||-(1) (H)_(3)C(H)_(2)C(H)_(2)C(O)_(2)Hxrightarrow ({(N{H)_(4))}_(2)C(O)_(3)}xrightarrow
-
2.完成下列反应式。-|||-(1) (H)_(3)C(H)_(2)C(H)_(2)C(O)_(2)Hxrightarrow ({(N{H)_(4))}_(2)
- 查看答案
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)^-1-|||-Ⅱ. (O)_(2)(g
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)
- 查看答案
-
路线如下:-|||-C2H5O-|||-Bn C3H3BrO 21 0 Bn-|||-C2H5O-|||-H B (D)-|||--C(H)_(3)cdot HCl H [N -C(H)_(3)-N-
-
路线如下:-|||-C2H5O-|||-Bn C3H3BrO 21 0 Bn-|||-C2H5O-|||-H B (D)-|||--C(H)_(3)cdot H
- 查看答案