=1.013times (10)^5Pa,-|||-_(1)=1330Pa , _(2)=7P(a)_(2),-|||-_(m)=1020kg/(h)_(2) , _(e)=0.755X,-|||-dfrac ({q)_(nl)}({q)_(nG)}=2(dfrac ({q)_(nt)}({q)_(n)G})_(min)-|||-therefore (y)_(1)=dfrac ({P)_(1)}(P)=dfrac (1330)(1.013times {10)^5}=0.01313-|||-_(2)=dfrac ({P)_(2)}(P)=dfrac (7)(1.013times {10)^5}=6.910times (10)^-5-|||-_(1)=dfrac ({y)_(1)}(1-{y)_(1)}=0.0133-|||-_(2)=dfrac ({y)_(2)}(1-{y)_(2)}=6.910times (10)^-5-|||-_(n)c=dfrac ({Q)_(m)v}(M)=dfrac ({Q)_(mn)}(Mg{y)_(1)+(M)_(min)}(1-(y)_(1)) _(n)((X)_(1)-(Y)_(2))=(a)_(n)((X)_(1)-(X)_(2))-|||-Itdan+24+100301 _(1)=dfrac ({G)_(mG)}({G)_(m)}((X)_(1)-(X)_(2))+(X)_(2)-|||-Itdan+24+100301-|||-(qnL )min=2·(1-√2)/(n-2)=2 Y1-Y2-,Y1-Y2 =dfrac (0.0133-6.910times {10)^-5}(1.502)+0-|||-m =8.809times (10)^-3-|||-m-|||-=2cdot dfrac (0.0133-6.910times {10)^-5}(0.0133/0.755-0)=1.502-|||-_(1)=(C)_(M)cdot (x)_(1)=dfrac ({P)_(s)}(M,)cdot dfrac ({X)_(1)}(1+{X)_(1)}-|||-_(n)=1.502(a)_(n6)=1.502times G(1-(y)_(1))-|||-=dfrac (998.2)(18)cdot dfrac (8.809times {10)^-3}(1+8.809times {10)^-3}-|||-=51.72kmol/h=930.96kg/h-|||-=0.4842kmol(m)^3_(2)=0, . =1.013times (10)^5Pa,-|||-_(1)=1330Pa , _(2)=7P(a)_(2),-|||-_(m)=1020kg/(h)_(2) , _(e)=0.755X,-|||-dfrac ({q)_(nl)}({q)_(nG)}=2(dfrac ({q)_(nt)}({q)_(n)G})_(min)-|||-therefore (y)_(1)=dfrac ({P)_(1)}(P)=dfrac (1330)(1.013times {10)^5}=0.01313-|||-_(2)=dfrac ({P)_(2)}(P)=dfrac (7)(1.013times {10)^5}=6.910times (10)^-5-|||-_(1)=dfrac ({y)_(1)}(1-{y)_(1)}=0.0133-|||-_(2)=dfrac ({y)_(2)}(1-{y)_(2)}=6.910times (10)^-5-|||-_(n)c=dfrac ({Q)_(m)v}(M)=dfrac ({Q)_(mn)}(Mg{y)_(1)+(M)_(min)}(1-(y)_(1)) _(n)((X)_(1)-(Y)_(2))=(a)_(n)((X)_(1)-(X)_(2))-|||-Itdan+24+100301 _(1)=dfrac ({G)_(mG)}({G)_(m)}((X)_(1)-(X)_(2))+(X)_(2)-|||-Itdan+24+100301-|||-(qnL )min=2·(1-√2)/(n-2)=2 Y1-Y2-,Y1-Y2 =dfrac (0.0133-6.910times {10)^-5}(1.502)+0-|||-m =8.809times (10)^-3-|||-m-|||-=2cdot dfrac (0.0133-6.910times {10)^-5}(0.0133/0.755-0)=1.502-|||-_(1)=(C)_(M)cdot (x)_(1)=dfrac ({P)_(s)}(M,)cdot dfrac ({X)_(1)}(1+{X)_(1)}-|||-_(n)=1.502(a)_(n6)=1.502times G(1-(y)_(1))-|||-=dfrac (998.2)(18)cdot dfrac (8.809times {10)^-3}(1+8.809times {10)^-3}-|||-=51.72kmol/h=930.96kg/h-|||-=0.4842kmol(m)^3
吸收:
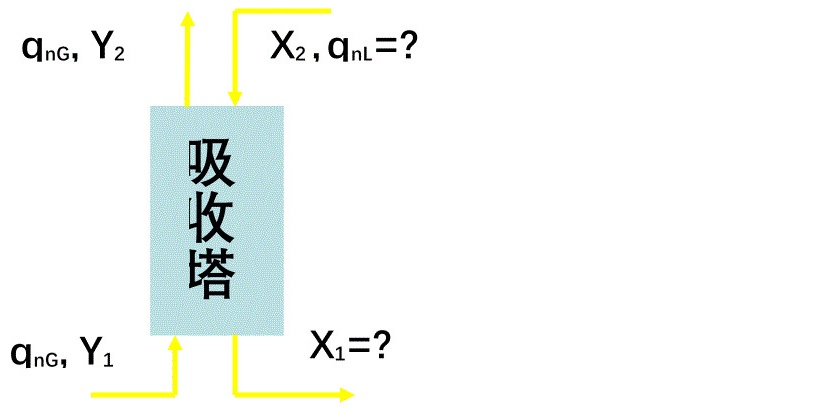
例1、在 20℃,1atm下,用清水分离氨-空气的混合气体,混合气体中氨的分压为 1330Pa,经吸收后氨的分压降为 7 Pa,混合气体的处理量为 1020 kg/h,操作条件下平衡关系为 Y = 0.755X。若适宜的吸收剂用量为最小用量的2倍,求所需吸收剂用量及离塔氨水的浓度。

参考答案与解析:
-
相关试题
-
经历下述可逆等压过程:_(1)=1.013times (10)^5Pa-|||-_(1)=3(dm)^3 可逆等压 _(2)=1.013times (10)^5Pa-|||-_(2)=5.25(dm)
-
经历下述可逆等压过程:_(1)=1.013times (10)^5Pa-|||-_(1)=3(dm)^3 可逆等压 _(2)=1.013times (10)^5
- 查看答案
-
12.9 容器内贮有氧气,其压强 =1.013times (10)^5Pa, 温度 t=27℃, 求:(1)单位体积内-|||-的分子数;(2)分子质量m0;(3)氧气的密度p;(4)方均根速率;(5
-
12.9 容器内贮有氧气,其压强 =1.013times (10)^5Pa, 温度 t=27℃, 求:(1)单位体积内-|||-的分子数;(2)分子质量m0;(
- 查看答案
-
有1mol的CO2,作如图所示的循环过程,ca为等温过程其中_(1)=2.0times (10)^5Pa,_(1)=2.0times (10)^5Pa,_(1)=2.0times (10)^5Pa求(
-
有1mol的CO2,作如图所示的循环过程,ca为等温过程其中_(1)=2.0times (10)^5Pa,_(1)=2.0times (10)^5Pa,_(1)
- 查看答案
-
/((10)^5Pa)-|||-4 A D-|||-3-|||-2-|||-1 E B-|||-2 3 4 V/(10^-3)m^3)
-
/((10)^5Pa)-|||-4 A D-|||-3-|||-2-|||-1 E B-|||-2 3 4 V/(10^-3)m^3)
- 查看答案
-
例7.4 一容器内贮有氧气,其压强为 .0atan (1atan m=1.013times (10)^5Pa), 温度为-|||-27℃,求:(1)单位体积的分子数。(2)氧气的密度。(3)氧分子的质
-
例7.4 一容器内贮有氧气,其压强为 .0atan (1atan m=1.013times (10)^5Pa), 温度为-|||-27℃,求:(1)单位体积的分
- 查看答案
-
习题16 假设N2为理想气体。在0C和 times (10)^5Pa 下,用2 dm^3 N 2作定温膨胀到压力-|||-为10^5Pa。-|||-(1)如果是可逆膨胀;-|||-(2)如果膨胀是在外
-
习题16 假设N2为理想气体。在0C和 times (10)^5Pa 下,用2 dm^3 N 2作定温膨胀到压力-|||-为10^5Pa。-|||-(1)如果是
- 查看答案
-
习题16 假设N2为理想气体。在0℃和 times (10)^5cdot (P)_(a) 下,用2 dm^3N2作定温膨胀到压力-|||-为10^5Pa。-|||-(1)如果是可逆膨胀;-|||-(2
-
习题16 假设N2为理想气体。在0℃和 times (10)^5cdot (P)_(a) 下,用2 dm^3N2作定温膨胀到压力-|||-为10^5Pa。-||
- 查看答案
-
习题34 假设He为理想气体。1 mol He 由 times (10)^5Pa 0℃变为10^5Pa、50℃,可经两个-|||-不同的途径:(1)先定压加热,再定温可逆膨胀;(2)先定温可逆膨胀;再
-
习题34 假设He为理想气体。1 mol He 由 times (10)^5Pa 0℃变为10^5Pa、50℃,可经两个-|||-不同的途径:(1)先定压加热,
- 查看答案
-
↑p/10^5Pa-|||-4 A-|||-D-|||-3-|||-2 C-|||-1 B-|||-0 1 2 3 4 V/(10^-3m^3)
-
↑p/10^5Pa-|||-4 A-|||-D-|||-3-|||-2 C-|||-1 B-|||-0 1 2 3 4 V/(10^-3m^3)
- 查看答案
-
5.20 有25 mo1的单原子气体,做如图5.24所示的循环-|||-过程(ac为等温过程)。 _(1)=4.15times (10)^5Pa _(1)=2.0times (10)^-2(m)^3,
-
5.20 有25 mo1的单原子气体,做如图5.24所示的循环-|||-过程(ac为等温过程)。 _(1)=4.15times (10)^5Pa _(1)=2.
- 查看答案