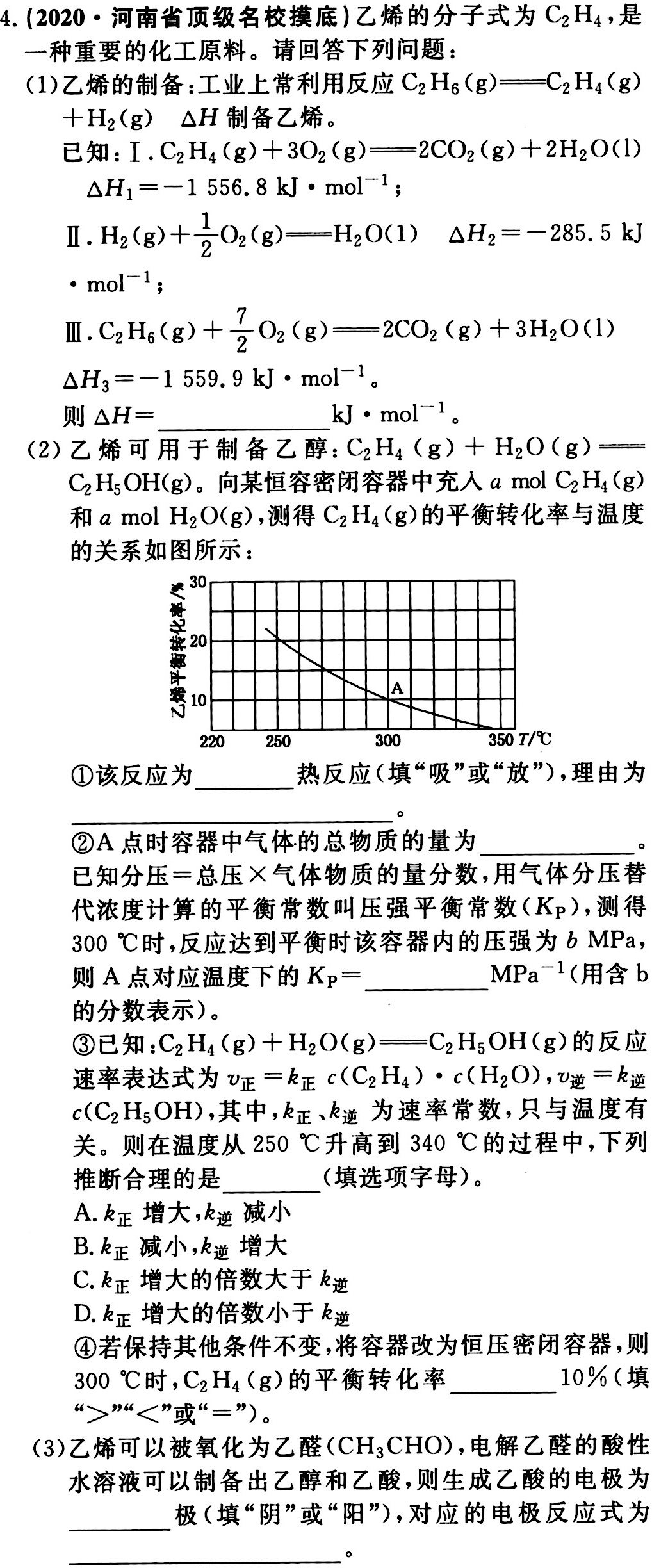
_(2)(g)+dfrac (1)(2)(O)_(2)(g)=!=!= (H)_(2)O(1) Delta (H)_(2)=-285.5kJ-|||-cdot (mol)^-1;-|||-Ⅲ. _(2)(H)_(6)(g)+dfrac (7)(2)(O)_(2)(g)=!=!= 2C(O)_(2)(g)+3(H)_(2)O(l)-|||-Delta (H)_(3)=-1559.9kJcdot (mol)^-1-|||-则 Delta H= __ cdot (mol)^-1-|||-__-|||-(2)乙烯可用于制备乙醇: _(2)(H)_(4)(g)+(H)_(2)O(g)=!=!= -|||-__-|||-C2H5OH(g)。向某恒容密闭容器中充入a mol C2H4(g)-|||-和amolH2O(g),测得C2H4(g)的平衡转化率与温度-|||-的关系如图所示:-|||-30-|||-20-|||-10 A-|||-220 250 300 350T/℃-|||-①该反应为 __ 热反应(填"吸"或"放"),理由为-|||-__-|||-②A点时容器中气体的总物质的量为 __-|||-已知分压=总压×气体物质的量分数,用气体分压替-|||-代浓度计算的平衡常数叫压强平衡常数(Kp),测得-|||-300℃时,反应达到平衡时该容器内的压强为bMPa,-|||-则A点对应温度下的 _(p)= __ (Pa)^-1 (用含b-|||-的分数表示)。-|||-③已知: _(2)(H)_(4)(g)+(H)_(2)O(g)=!=!= (C)_(2)(H)_(5)OH(g) 的反应-|||-速率表达式为 _(正)=(k)_(正)c((C)_(2)(H)_(4))cdot c((H)_(2)O), _(UND)=(k)_(UND)-|||-c(C2H5OH),其中,k正、k逆为速率常数,只与温度有-|||-关。则在温度从250℃升高到340℃的过程中,下列-|||-推断合理的是 __ (填选项字母)。-|||-A.k正增大,k逆减小-|||-B.k正减小,k逆增大-|||-C.k正增大的倍数大于k逆-|||-D.k正增大的倍数小于k逆-|||-④若保持其他条件不变,将容器改为恒压密闭容器,则-|||-300℃时,C2H4(g)的平衡转化率 __ 10%(填-|||-">""
参考答案与解析:
-
相关试题
-
已知:( bigcirc (1)C(H)_(5)OH(g)+dfrac (1)(2)(O)_(2)(g)=C(O)_(2)(g)+2(H)_(2)(g) Delta H=-192.9kJ/mol-|
-
已知:( bigcirc (1)C(H)_(5)OH(g)+dfrac (1)(2)(O)_(2)(g)=C(O)_(2)(g)+2(H)_(2)(g) De
- 查看答案
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+(H)_(2)(g)-|||-Delta
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+
- 查看答案
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)^-1-|||-Ⅱ. (O)_(2)(g
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)
- 查看答案
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol) -235.3 52.28 -241.
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol
- 查看答案
-
298.15K时,反应 (H)_(2)(O)_(2)(1)leftharpoons 2(H)_(2)O(1)+(O)_(2)(g) 的 (Delta )_(r)(H)_(m)^theta =-|||-
-
298.15K时,反应 (H)_(2)(O)_(2)(1)leftharpoons 2(H)_(2)O(1)+(O)_(2)(g) 的 (Delta )_(r)
- 查看答案
-
(H)_(2)(g)+(O)_(2)(g)=!=!= 2(H)_(2)O(l) Delta (H)_(3)=-572kJcdot mo(l)^-1-|||-_(2)S(g)=!=!= S(s)+(H
-
(H)_(2)(g)+(O)_(2)(g)=!=!= 2(H)_(2)O(l) Delta (H)_(3)=-572kJcdot mo(l)^-1-|||-_
- 查看答案
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ. _(2)(H)_(5)OH(g)=C
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ
- 查看答案
-
18.已知:-|||-①H2O(g)=H2(g)+2O2(g) H=+241.88kJ/mol-|||-②C(s)+2O2(g )=CO(g) △H=-110.50.5kJ/mol-|||-③C(s)
-
18.已知:-|||-①H2O(g)=H2(g)+2O2(g) H=+241.88kJ/mol-|||-②C(s)+2O2(g )=CO(g) △H=-110.
- 查看答案
-
对于反应N2(g)+O2(g)2NO(g),△H=+90kJ/moL,△S=+1
-
[单选题]对于反应N2(g)+O2(g)2NO(g),△H=+90kJ/moL,△S=+12J/(K·mol),下列哪种情况是正确的?()A . 任何温度下均自发B . 任何温度下均非自发C . 低温下非自发,高温下自发D . 低温下自发,高温下非自发
- 查看答案
-
在298K时,H2(g)+1/2O2(g)=H2O(L),△H=-285.8kJ
-
[单选题]在298K时,H2(g)+1/2O2(g)=H2O(L),△H=-285.8kJ/mol。若温度升高,则有下列中何种变化?()A . 正反应速率增大,逆反应速率减小B . 正反应速率增大,逆反应速率增大C . 正反应速率减小,逆反应速率增大D . 正反应速率减小,逆反应速率减小
- 查看答案