在1000K,反应_(z)+2(H)_(2)(g)=C(H)_(4)(g)的标准摩尔反应焓_(z)+2(H)_(2)(g)=C(H)_(4)(g)与温度无关,_(z)+2(H)_(2)(g)=C(H)_(4)(g)(1) 该反应在1200K、500kPa下,由H2(g)与C(石)开始反应,平衡时系统中H2(g)的分压为多少?(2) 在上述平衡系统中,恒温恒容下充入惰性气体,平衡将如何移动?为什么?
在1000K,反应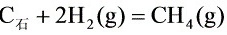 的标准摩尔反应焓
的标准摩尔反应焓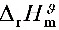 与温度无关,
与温度无关,
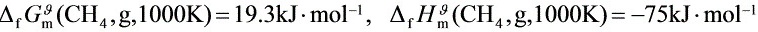
(1) 该反应在1200K、500kPa下,由H2(g)与C(石)开始反应,平衡时系统中H2(g)的分压为
多少?
(2) 在上述平衡系统中,恒温恒容下充入惰性气体,平衡将如何移动?为什么?
参考答案与解析:
-
相关试题
-
用键焓来估算反应热时,反应_(2)(H)_(6)(g)arrow (C)_(2)(H)_(4)(g)+(H)_(2)(g)的热效应_(2)(H)_(6)(g)arrow (C)_(2)(H)_(4)(
-
用键焓来估算反应热时,反应_(2)(H)_(6)(g)arrow (C)_(2)(H)_(4)(g)+(H)_(2)(g)的热效应_(2)(H)_(6)(g)a
- 查看答案
-
2.13 已知下列反应在298K时的标准摩尔焓变:-|||-(1) (C)_(2)(H)_(2)(g)+5(O)_(2)(g)=4C(O)_(2)(g)+2(H)_(2)O(g) (Delta )_(
-
2.13 已知下列反应在298K时的标准摩尔焓变:-|||-(1) (C)_(2)(H)_(2)(g)+5(O)_(2)(g)=4C(O)_(2)(g)+2(H
- 查看答案
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ. _(2)(H)_(5)OH(g)=C
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ
- 查看答案
-
对于反应 (H)_(4)(g)+2(O)_(2)(g)=!=!= C(O)_(2)(g)+2(H)_(2)O(g)-|||-(1)在恒压下,升高反应体系的温度,此反应的标准平衡常数K^2/ () ,C
-
对于反应 (H)_(4)(g)+2(O)_(2)(g)=!=!= C(O)_(2)(g)+2(H)_(2)O(g)-|||-(1)在恒压下,升高反应体系的温度,
- 查看答案
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)^-1-|||-Ⅱ. (O)_(2)(g
-
_(2)(H)_(6)(g)=!=!= (C)_(2)(H)_(4)(g)+(H)_(2)(g) Delta (H)_(1)=+137kJcdot (mol)
- 查看答案
-
(rm C(g)+H_2O(g) {!=!=!=) CO(g)+H_2(g)} Delta H_2iii.(rm C(s)+2H_2(g) {!=!=!=) CH_4(g)} Delt
-
(rm C(g)+H_2O(g) {!=!=!=) CO(g)+H_2(g)} Delta H_2iii.(rm C(s)+2H_2(g) {!=!=!
- 查看答案
-
已知反应C2H2(g)+2H2(g)C2H6(g)的,当反应达到平衡后,欲使反应
-
[单选题]已知反应C2H2(g)+2H2(g)C2H6(g)的,当反应达到平衡后,欲使反应向右进行,可采取的方法是()。A . 升温,升压B . 升温,减压C . 降温,升压D . 降温,减压
- 查看答案
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol) -235.3 52.28 -241.
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol
- 查看答案
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+(H)_(2)(g)-|||-Delta
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+
- 查看答案
-
(2013)已知反应C2H2(g)+2H2(g)C2H6(g)的△rHm<0,当
-
[单选题](2013)已知反应C2H2(g)+2H2(g)C2H6(g)的△rHm<0,当反应达平衡后,欲使反应向右进行,可采取的方法是:()A . 升温,升压B . 升温,减压C . 降温,升压D . 降温,减压
- 查看答案