(4) (s)+CuS(O)_(4)(aq)-|||-11.计算下列反应的:(1)△,H^(29^2)(15K);(2)△,U 298.15K)和(3)298.15K进行-|||-反应进度为1 mol的体积功。-|||-(H)_(4)(g)+4(Cl)_(2)(g)=!=!= C(Cl)_(4)(1)+4HCl(g) __
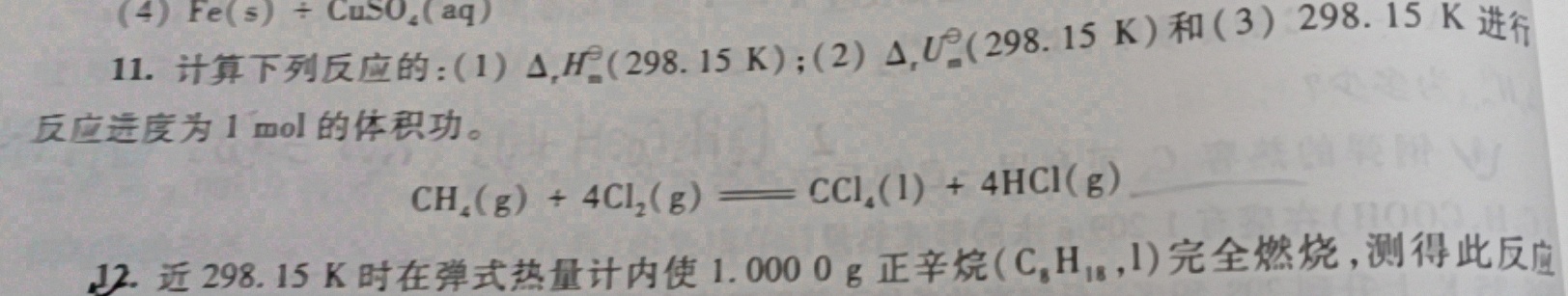
参考答案与解析:
-
相关试题
-
11.计算下列反应的:(1) (Delta )_(1)(H)_(m)(298.15k) ;(2) Delta (U)_(m)(298.15k) 和(3)298.15K进行-|||-反应进度为1 mol
-
11.计算下列反应的:(1) (Delta )_(1)(H)_(m)(298.15k) ;(2) Delta (U)_(m)(298.15k) 和(3)298.
- 查看答案
-
已知MgCO3(s)的ΔfHm (298.15K) =-1111.88kJ.mol-1,Sm (298.15K)=65.6J.K-1mol-1,ΔfGm (298.15K) =-1028.28k
-
已知MgCO3(s)的ΔfHm (298.15K) =-1111.88kJ.mol-1,Sm (298.15K)=65.6J.K-1mol-1,ΔfGm
- 查看答案
-
2-11 已知下列反应在298.15 K的平衡常数,计算反应 2CO(g)-|||-(s)+2C(O)_(2)(g) 在298.15K时的平衡常数K。-|||-(1) (O)_(2)(s)+2(H)_
-
2-11 已知下列反应在298.15 K的平衡常数,计算反应 2CO(g)-|||-(s)+2C(O)_(2)(g) 在298.15K时的平衡常数K。-|||-
- 查看答案
-
山风-|||-10.查阅附录3的数据,试计算下列反应的 (Delta )_(1)(H)_(m)(298.15k)-|||-(1) (H)_(3)(g)+3(O)_(2)(g)=!=!= 2(N)_(2
-
山风-|||-10.查阅附录3的数据,试计算下列反应的 (Delta )_(1)(H)_(m)(298.15k)-|||-(1) (H)_(3)(g)+3(O)
- 查看答案
-
试判断下列反应:N2 (g) + 3 H2 (g) ® 2 NH 3 (g) (1) 在298.15K,标准态下能否自发进行? (2) 计算298.15K时该反应的平衡常数Kθ值。 (3) 欲使上述反
-
试判断下列反应:N2 (g) + 3 H2 (g) ® 2 NH 3 (g) (1) 在298.15K,标准态下能否自发进行? (2) 计算298.15K时该反
- 查看答案
-
298.15K时,反应 (H)_(2)(O)_(2)(1)leftharpoons 2(H)_(2)O(1)+(O)_(2)(g) 的 (Delta )_(r)(H)_(m)^theta =-|||-
-
298.15K时,反应 (H)_(2)(O)_(2)(1)leftharpoons 2(H)_(2)O(1)+(O)_(2)(g) 的 (Delta )_(r)
- 查看答案
-
15.已知反应:-|||-.dfrac (1)(2)(H)_(2)(g)+dfrac (1)(2)(Cl)_(2)(g)leftharpoons HCl(g)-|||-在298.15K时的 _(1)=
-
15.已知反应:-|||-.dfrac (1)(2)(H)_(2)(g)+dfrac (1)(2)(Cl)_(2)(g)leftharpoons HCl(g)-
- 查看答案
-
298.15K时,反应(H)_(2)(O)_(2) (l) leftharpoons 2(H)_(2) O (l) +(O)_(2)(g)的(H)_(2)(O)_(2) (l) leftharpoon
-
298.15K时,反应(H)_(2)(O)_(2) (l) leftharpoons 2(H)_(2) O (l) +(O)_(2)(g)的(H)_(2)(O)
- 查看答案
-
298.15K时反应 (O)_(2)(g)+dfrac (1)(2)(O)_(2)(g)=S(O)_(3)(g) 的 ^erasure =2.6times (10)^12, 标准摩尔反应焓 ({Del
-
298.15K时反应 (O)_(2)(g)+dfrac (1)(2)(O)_(2)(g)=S(O)_(3)(g) 的 ^erasure =2.6times (1
- 查看答案
-
15.已知反应:-|||-dfrac (1)(2)(H)_(2)(g)+dfrac (1)(2)(Cl)_(2)(g)leftharpoons HCl(g)-|||-在298.15K时的 _(1)^t
-
15.已知反应:-|||-dfrac (1)(2)(H)_(2)(g)+dfrac (1)(2)(Cl)_(2)(g)leftharpoons HCl(g)-|
- 查看答案