4,处在热力学温度为T的平衡态的1 mol氦气(视为单原子分子理想气体)的内能为
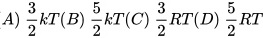
(k为玻尔兹曼常数,R为普适气体常数)
4,处在热力学温度为T的平衡态的1 mol氦气(视为单原子分子理想气体)的内能为
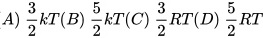
(k为玻尔兹曼常数,R为普适气体常数)
3,处在热力学温度为了的平衡态的单原子分子理想气体,分子的平均平动动能为( ) (k为玻尔兹曼常数,R为普适气体常数)(A) dfrac (5)(2)kJ (B
对于处在平衡态下温度为T的理想气体,dfrac (3)(2)kT的物理意义是________________________________________.(
dfrac (3)(2)RC dfrac (7)(2)R D. dfrac (3)(2)RT对于双原子气体的摩尔定压热容的表达式为()
试说明下列各量的物理意义。(1)(1)/(2)kT(2)(3)/(2)kT(3)(i)/(2)kT(4)(M)/(M_(mol))(i)/(2)RT(5)(i)
单原子分子理想气体的定容摩尔热容: ( )A.dfrac (1)(2)RB.dfrac (1)(2)RC.dfrac (1)(2)R
dfrac (1)(5)(X)_(1)+dfrac (2)(5)(X)_(2)+dfrac (2)(5)(X)_(3)-|||-D .dfrac (1)(7)(
B. dfrac {1)(2) . C. dfrac (2)(3) . D. dfrac (3)(4)
(B) =-dfrac (x)(2)+dfrac (3)(2)-|||-(C) =dfrac (x)(2)+dfrac (3)(2) (D) =-dfrac (
已知_(1)=((dfrac {1)(3),-dfrac (2)(3),-dfrac (2)(3))}^T, _(2)=((-dfrac {2)(3),dfra
(B) =2, =dfrac (1)(3).-|||-(C) =1, =dfrac (1)(2). (D) =1, =-dfrac (1)(3),求指导本题解题