氢能是一种极具发展潜力的清洁能源。下列反应是目前大规模制取氢气的方法之一、(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)(1)在生产中,欲使CO的转化率提高,同时提高(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)的产率,可采取哪些措施___________?(2)在容积不变的密闭容器中,将2.0molCO与8.0mol(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)混合加热到830℃发生上述反应,达到平衡时CO的转化率是80%。计算该反应的平衡常数___________。(3)实验发现,其他条件不变,在相同时间内,向上述体系中投入一定量的CaO可以明显提高(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)的体积分数。对比实验的结果如图所示。(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)请思考:投入CaO时,(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)的体积分数为什么会增大___________?微米CaO和纳米CaO对平衡的影响为何不同___________?
氢能是一种极具发展潜力的清洁能源。下列反应是目前大规模制取氢气的方法之一、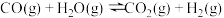
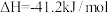
(1)在生产中,欲使CO的转化率提高,同时提高
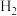 的产率,可采取哪些措施
的产率,可采取哪些措施(2)在容积不变的密闭容器中,将2.0molCO与8.0mol
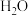 混合加热到830℃发生上述反应,达到平衡时CO的转化率是80%。计算该反应的平衡常数
混合加热到830℃发生上述反应,达到平衡时CO的转化率是80%。计算该反应的平衡常数(3)实验发现,其他条件不变,在相同时间内,向上述体系中投入一定量的CaO可以明显提高
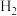 的体积分数。对比实验的结果如图所示。
的体积分数。对比实验的结果如图所示。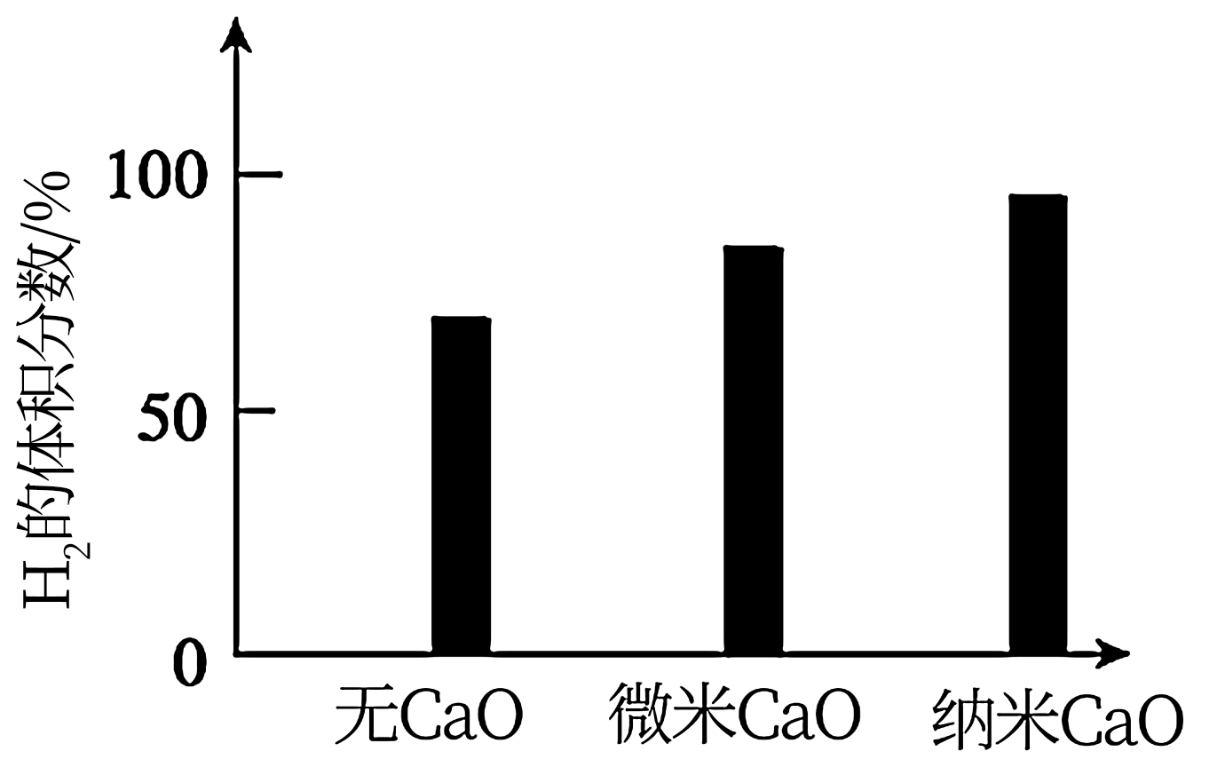
请思考:投入CaO时,
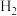 的体积分数为什么会增大
的体积分数为什么会增大
参考答案与解析:
-
相关试题
-
6.工业上制取氢气时涉及的一个重要反应是:-|||-(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)-|||-已知:-|||-①C(s)+1/2 O2(g)=CO
-
6.工业上制取氢气时涉及的一个重要反应是:-|||-(g)+(H)_(2)O(g)=!=!= C(O)_(2)(g)+(H)_(2)(g)-|||-已知:-||
- 查看答案
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+(H)_(2)(g)-|||-Delta
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+
- 查看答案
-
(g)+(H)_(2)O(g)leftharpoons (H)_(2)(g)+C(O)_(2)(g)-|||-某温度下达到平衡时-|||-(CO)=c((H)_(2)O)=0.005molcdot (
-
(g)+(H)_(2)O(g)leftharpoons (H)_(2)(g)+C(O)_(2)(g)-|||-某温度下达到平衡时-|||-(CO)=c((H)_
- 查看答案
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ. _(2)(H)_(5)OH(g)=C
-
_(2)(H)_(5)OH(g)+(H)_(2)O(g)=2(H)_(2)(g)+C(H)_(2)COOH(g) Delta (H)_(1) K-|||-Ⅱ
- 查看答案
-
6,水煤气发生炉中共有C(s),H 20(g),C 0(g),C 02(g),H 2(g)五种物质,其间能发生反应:CO 2(g)+C(s)=2 CO(g),H 2 O(g)+C(s)=CO(g)+H
-
6,水煤气发生炉中共有C(s),H 20(g),C 0(g),C 02(g),H 2(g)五种物质,其间能发生反应:CO 2(g)+C(s)=2 CO(g),H
- 查看答案
-
对于反应 (H)_(4)(g)+2(O)_(2)(g)=!=!= C(O)_(2)(g)+2(H)_(2)O(g)-|||-(1)在恒压下,升高反应体系的温度,此反应的标准平衡常数K^2/ () ,C
-
对于反应 (H)_(4)(g)+2(O)_(2)(g)=!=!= C(O)_(2)(g)+2(H)_(2)O(g)-|||-(1)在恒压下,升高反应体系的温度,
- 查看答案
-
已知:( bigcirc (1)C(H)_(5)OH(g)+dfrac (1)(2)(O)_(2)(g)=C(O)_(2)(g)+2(H)_(2)(g) Delta H=-192.9kJ/mol-|
-
已知:( bigcirc (1)C(H)_(5)OH(g)+dfrac (1)(2)(O)_(2)(g)=C(O)_(2)(g)+2(H)_(2)(g) De
- 查看答案
-
已知某温度下,下列可逆反应的标准平衡常数:-|||-(H)_(2)(g)+(O)_(2)(g)leftharpoons 2(H)_(2)O(g) K-|||-(g)+(O)_(2)(g)leftha
-
已知某温度下,下列可逆反应的标准平衡常数:-|||-(H)_(2)(g)+(O)_(2)(g)leftharpoons 2(H)_(2)O(g) K-|||-
- 查看答案
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol) -235.3 52.28 -241.
-
判断反应 _(2)(H)_(5)OH(g)=(C)_(2)(H)_(4)(g)+(H)_(2)O(g)-|||-已知 Delta (H)_(m)O(kJ/mol
- 查看答案
-
18.已知:-|||-①H2O(g)=H2(g)+2O2(g) H=+241.88kJ/mol-|||-②C(s)+2O2(g )=CO(g) △H=-110.50.5kJ/mol-|||-③C(s)
-
18.已知:-|||-①H2O(g)=H2(g)+2O2(g) H=+241.88kJ/mol-|||-②C(s)+2O2(g )=CO(g) △H=-110.
- 查看答案