用公式Delta H(T)=Delta H(298)+(int )_(Delta )(C)_(p)dT计算某反应在温度T的反应焓变,要求:
用公式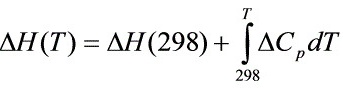 计算某反应在温度T的反应焓变,要求:
计算某反应在温度T的反应焓变,要求:
B. T>298K;
C. △CP≠0;
D. 298~T间反应物和产物无相变
参考答案与解析:
-
相关试题
-
17.化学反应的标准摩尔反应焓随温度的变化率 dfrac (d{{Delta )_(1)}^theta }(dT)= () ,在一-|||-定的温度范围内标准摩尔反应焓 Delta (H)_(m)^t
-
17.化学反应的标准摩尔反应焓随温度的变化率 dfrac (d{{Delta )_(1)}^theta }(dT)= () ,在一-|||-定的温度范围内标准摩
- 查看答案
-
11.某化学反应的 Delta (H)_(298)lt 0 , Delta (S)_(298)gt 0 ,在298K时该反应有: ()-|||-
-
11.某化学反应的 Delta (H)_(298)lt 0 , Delta (S)_(298)gt 0 ,在298K时该反应有: ()-|||-
- 查看答案
-
(B) (int )_(x)^x+Delta xf(t)dtlt f(x)Delta xlt 0.-|||-(C) (x)Delta xgt (int )_(x)^x+Delta xf(t)dtgt
-
(B) (int )_(x)^x+Delta xf(t)dtlt f(x)Delta xlt 0.-|||-(C) (x)Delta xgt (int )_(x
- 查看答案
-
(B) (int )_(x)^x+Delta xf(t)dtlt f(x)Delta xlt 0.-|||-(C) (x)Delta xgt (int )_(x)^x+Delta xf(t)dtgt
-
(B) (int )_(x)^x+Delta xf(t)dtlt f(x)Delta xlt 0.-|||-(C) (x)Delta xgt (int )_(x
- 查看答案
-
在等压过程中,气体吸收的热量公式为:=v(C)_(v)Delta T=v(C)_(v)Delta T=v(C)_(v)Delta T=v(C)_(v)Delta T
-
在等压过程中,气体吸收的热量公式为:=v(C)_(v)Delta T=v(C)_(v)Delta T=v(C)_(v)Delta T=v(C)_(v)Delta
- 查看答案
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+(H)_(2)(g)-|||-Delta
-
Delta (H)_(1)=akJcdot (mol)^-1-|||-反应Ⅱ: (g)+(H)_(2)O(g)leftharpoons C(O)_(2)(g)+
- 查看答案
-
2.13 已知下列反应在298K时的标准摩尔焓变:-|||-(1) (C)_(2)(H)_(2)(g)+5(O)_(2)(g)=4C(O)_(2)(g)+2(H)_(2)O(g) (Delta )_(
-
2.13 已知下列反应在298K时的标准摩尔焓变:-|||-(1) (C)_(2)(H)_(2)(g)+5(O)_(2)(g)=4C(O)_(2)(g)+2(H
- 查看答案
-
2、反应NH4HS(s )已 )+(H)_(2)S(g) 的 Delta (H)_(m)(298K)=93.72kJcdot mo(l)^-1, 设 Delta (H)_(m)^theta (m)-|
-
2、反应NH4HS(s )已 )+(H)_(2)S(g) 的 Delta (H)_(m)(298K)=93.72kJcdot mo(l)^-1, 设 Delta
- 查看答案
-
Delta H Delta S Delta A 及 Delta G-|||-[答案: =-8.31times (10)^3J =8.31times (10)^3J Delta U=Delta H=0;
-
Delta H Delta S Delta A 及 Delta G-|||-[答案: =-8.31times (10)^3J =8.31times (10)^3
- 查看答案