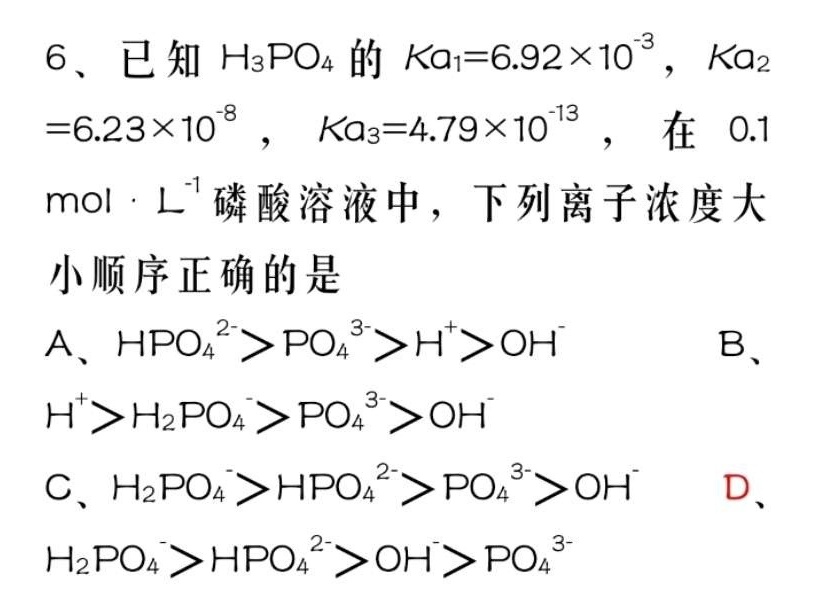
6、已知H3PO4的 (a)_(1)=6.92times (10)^-3 Ka2-|||-=6.23times (10)^-8 , _({a)_(3)}=4.79times (10)^-13, 在0.1-|||-cdot (L)^-1 磷酸溶液中,下列离子浓度大-|||-小顺序正确的是-|||-A、 (O)_(4)^2-gt (P{O)_(4)}^3-gt (H)^+gt (OH)^- B、-|||-^+gt (H)_(2)P(O)_(4)gt (P{O)_(4)}^3gt (OH)^--|||-C、 _(2)P(O)_(4)gt HP(O)_(4)^2-gt (P{O)_(4)}^3-gt (OH)^- D、-|||-_(2)P(O)_(4)gt HP(O)_(4)^2-gt OHgt (P{O)_(4)}^3-
参考答案与解析:
-
相关试题
-
用NaOH滴定mathrm(H)_3mathrm(AsO)_4(mathrm(Ka)_1=6.5times10^-3, mathrm(Ka)_2=1.1times10^-7, mathrm(Ka)_3
-
用NaOH滴定mathrm(H)_3mathrm(AsO)_4(mathrm(Ka)_1=6.5times10^-3, mathrm(Ka)_2=1.1time
- 查看答案
-
9-17 如题图 9-17 所示,已知 =8times (10)^-2m,-|||-=6times (10)^-2m, _(1)=3times (10)^-8C, _(2)=-3times (10)^
-
9-17 如题图 9-17 所示,已知 =8times (10)^-2m,-|||-=6times (10)^-2m, _(1)=3times (10)^-8C
- 查看答案
-
计算: (1)((-1))^10times 2+((-2))^3div 4; (2)((-1))^10times 2+((-2))^3div 4; (3)((-1))^10times 2+((-2))
-
计算: (1)((-1))^10times 2+((-2))^3div 4; (2)((-1))^10times 2+((-2))^3div 4; (3)((-
- 查看答案
-
以 NaOH 滴定 H3PO4(Ka1=7.5×10−3;Ka2=6.2×10−8;Ka3=5.0×10−13) 至生成 NaH2PO4 时溶液的 pH 值为( )A.2.3B.2.6C.3.6
-
以 NaOH 滴定 H3PO4(Ka1=7.5×10−3;Ka2=6.2×10−8;Ka3=5.0×10−13) 至生成 NaH2PO4 时溶液的 pH 值为(
- 查看答案
-
13.已知H2 CO3的 _(o1)((H)_(2)C(O)_(3))=4.2times (10)^-7 _(a2)((H)_(2)C(O)_(3))=4.8times (10)^-11, 则 1.0
-
13.已知H2 CO3的 _(o1)((H)_(2)C(O)_(3))=4.2times (10)^-7 _(a2)((H)_(2)C(O)_(3))=4.8t
- 查看答案
-
如图所示,已知 r=6cm d=8cm, _(1)=3times (10)^-8C, _(2)=-3times (10)^-8C, 求(1)将电荷量为-|||-times (10)^-9C 的点电荷从
-
如图所示,已知 r=6cm d=8cm, _(1)=3times (10)^-8C, _(2)=-3times (10)^-8C, 求(1)将电荷量为-|||-
- 查看答案
-
11.已知:-|||-酸 H2C2O4 HF H2CO3 H3BO3-|||-电离平 _({a)_(1)}=5.9times (10)^-2 _({B)_(1)}=4.2times (10)^-7-|
-
11.已知:-|||-酸 H2C2O4 HF H2CO3 H3BO3-|||-电离平 _({a)_(1)}=5.9times (10)^-2 _({B)_(1)
- 查看答案
-
已知H3PO4的K1=7.6×10-3,K2=6.8×10-8,K3=4.4×1
-
[填空题] 已知H3PO4的K1=7.6×10-3,K2=6.8×10-8,K3=4.4×10-13,则H2PO4-的共轭碱是(),H2PO4-的()Kb
- 查看答案
-
19.已知 (BaS(O)_(4))=1.07times (10)^-10 , (BaC(O)_(3))=2.58times (10)^-9 ,在1LNa2 CO3溶液中,转-|||-0.020mol
-
19.已知 (BaS(O)_(4))=1.07times (10)^-10 , (BaC(O)_(3))=2.58times (10)^-9 ,在1LNa2 C
- 查看答案
-
四个点电荷_(1)=(q)_(2)=(q)_(3)=(q)_(4)=1.0times (10)^-8C,分别放置在边长_(1)=(q)_(2)=(q)_(3)=(q)_(4)=1.0times (10
-
四个点电荷_(1)=(q)_(2)=(q)_(3)=(q)_(4)=1.0times (10)^-8C,分别放置在边长_(1)=(q)_(2)=(q)_(3)=
- 查看答案