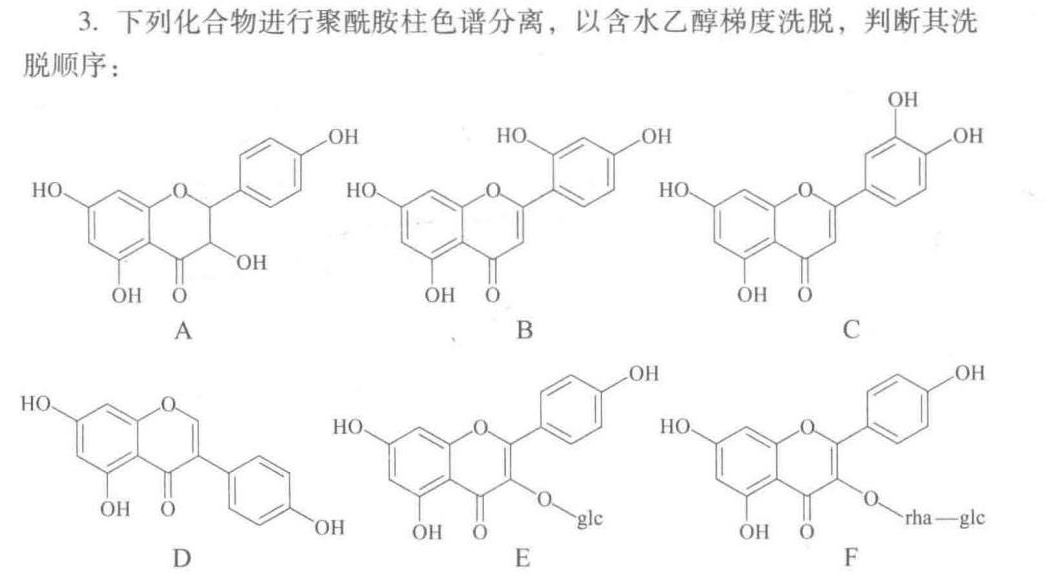
HO .0.-|||-OH-|||-OH o OH o OH o-|||-A B C-|||-OH OH-|||-HO 0-|||-HO 0 HO -0-|||-OH o 0. 0.-|||-OH OH o glc OH o-|||-rha -glc-|||-D E F
参考答案与解析:
-
相关试题
-
请比较下列物质酸水解反应的快慢:-|||-1)[填空1]>[填空2]>[填空3]-|||-OH-|||--OH H2 -OH-|||-CH2OH-|||-OH HO 一-|||-OH-||
-
请比较下列物质酸水解反应的快慢:-|||-1)[填空1]>[填空2]>[填空3]-|||-OH-|||--OH H2 -OH-|||-CH2OH-|||-OH
- 查看答案
-
10-8 完成下列转化:-|||-O COOH-|||-(1)-|||-0 O-|||-(2) (H)_(3)C-C(H)_(2)Br OH-|||-(3) → -CHO-|||-×(4) OH →
-
10-8 完成下列转化:-|||-O COOH-|||-(1)-|||-0 O-|||-(2) (H)_(3)C-C(H)_(2)Br OH-|||-(3) →
- 查看答案
-
H -OH HO H-|||-C2H5 CH3 CH3 CH3-|||-COOH COOH-|||-H OH HO H D. 与-|||-C. H -OH 与 H -OH-|||-COOH COOH
-
H -OH HO H-|||-C2H5 CH3 CH3 CH3-|||-COOH COOH-|||-H OH HO H D. 与-|||-C. H -OH 与
- 查看答案
-
聚乳酸是一种新型的生物可降解高分子材料,其合成路线如图:CH3 催化剂-|||-OH-|||-HO-|||-ǒ-|||-乳酸-|||-H OH H2O-|||-0 + m-|||-n-|||-聚乳酸下
-
聚乳酸是一种新型的生物可降解高分子材料,其合成路线如图:CH3 催化剂-|||-OH-|||-HO-|||-ǒ-|||-乳酸-|||-H OH H2O-|||-
- 查看答案
-
化合物F是治疗实体瘤的潜在药物。F的一条合成路线如图(略去部分试剂和条件);OH-|||-o-|||-OH-|||-Br2 C4H6Br2O 碱 o Br-|||-Br 碱 o-|||-A B C 中
-
化合物F是治疗实体瘤的潜在药物。F的一条合成路线如图(略去部分试剂和条件);OH-|||-o-|||-OH-|||-Br2 C4H6Br2O 碱 o Br-||
- 查看答案
-
(H)_(2)=CHC(H)_(2)OH-|||-OCH3-|||-CH3CH2CHCHCH3-|||-OH-|||-OH-|||-CH3CH2CHCHCH2 OH-|||-HQ CH3-|||-5.
-
(H)_(2)=CHC(H)_(2)OH-|||-OCH3-|||-CH3CH2CHCHCH3-|||-OH-|||-OH-|||-CH3CH2CHCHCH2
- 查看答案
-
NH4HS D (CH3CO)2O-|||-OH AlCl3 OH HCl OH H2O (C10H15NO)-|||-A B C-|||-段 (C4H10ClN H 1)HCl(浓) H (O)_(
-
NH4HS D (CH3CO)2O-|||-OH AlCl3 OH HCl OH H2O (C10H15NO)-|||-A B C-|||-段 (C4H10Cl
- 查看答案
-
equiv CC(H)_(2)C(H)_(2)OH 与 (H)_(3)C=!=!= C(C)_(2)OH-|||-CH2OH-|||-b. 与 CH3-|||-OH-|||-c.CH3CH2 OCH2
-
equiv CC(H)_(2)C(H)_(2)OH 与 (H)_(3)C=!=!= C(C)_(2)OH-|||-CH2OH-|||-b. 与 CH3-|||-
- 查看答案
-
25 ∘C 时,电极反应 ({{O)}_2}+2({{H)}^+}+2({{e)}^-}=({{H)}_2}({{O)}_2} 的标准电极电势为 0.(68 V),已知 φ⊖O2|OH−|H2O=0.
-
25 ∘C 时,电极反应 ({{O)}_2}+2({{H)}^+}+2({{e)}^-}=({{H)}_2}({{O)}_2} 的标准电极电势为 0.(68 V
- 查看答案